Silicone & Simulations
Tools to Replicate Clinical Conditions and Showcase Innovation
With more than 15 years of experience, BDC Laboratories is a global leader in engineered silicone vasculature. Using proprietary fabrication methods, we produce compliant mock vessels, anatomical silicone models, and full simulation systems for many of the world’s leading cardiovascular device companies.
Our Silicone and simulation solutions are used across the device lifecycle, from feasibility studies and design verification testing to regulatory evaluation, physician training, and product demonstrations. Whether you need simple vessel geometry, a patient-specific anatomical model, or a complete simulated use system that mimics physiological flow conditions, BDC Laboratories designs and delivers systems tailored to your requirements.
Anatomical Silicone Vessels →
Silicone Mock Vessels →
Simulation and Flow Models →
Training, Sales & Marketing Models →
Anatomical Additions →
Anatomical Silicone Vessels
Arterial Vasculature
Venous Vasculature
Healthy Vasculature
Diseased Vasculature
Silicone Mock Vessels
BDC Laboratories produces silicone mock vessels in diverse geometries. Vessels can be certified for geometry and either wall thickness or compliance, depending on the testing requirements.

Straight
Linear vessel segments for controlled evaluation of device performance across defined diameters and lengths.

U-Vessel
Curved geometries that replicate physiological bends to assess device functional performance and fatigue and durability in a challenging condition.

Bifurcated & Custom
Branching or complex anatomies, including patient-specific and disease-state models for advanced testing and training.
Simulation and Flow Models

Aortic Valve
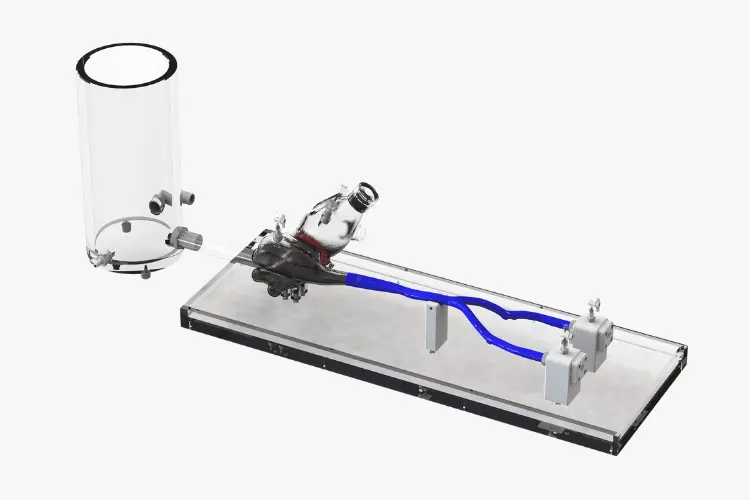
Mitral Valve

Tricuspid Valve
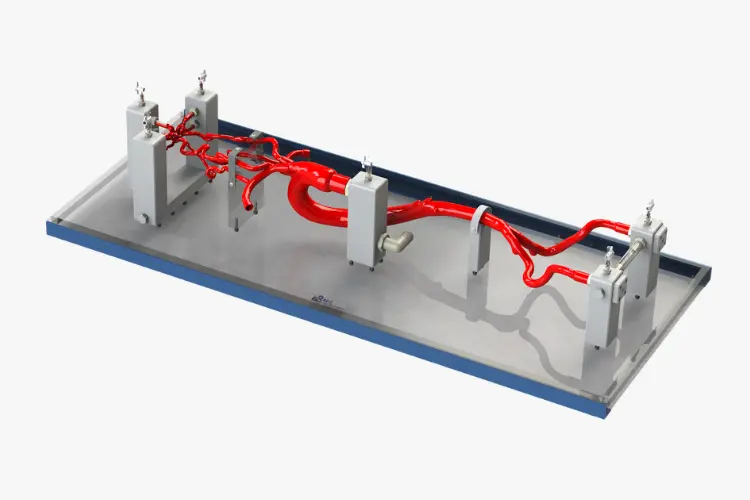
Neurovascular

Coronary Artery

Peripheral Leg

LAA / Septal Crossing

Physician Training Suites
Medical Training Models
BDC Laboratories provides a quick, effective way to help healthcare professionals understand how to use your cardiac, endovascular, and vascular medical devices with custom anatomical models that are clinically relevant, portable, and include the anatomy you need and nothing else.
Anatomical Additions
Optional technologies are available to increase model fidelity and tailor systems to specific clinical use cases. These technologies support more realistic simulation and device interaction.
SLIC™ Friction Reduction Coating
BDC Laboratories’ proprietary SLIC™ Friction Reduction Coating is engineered to provide a clinically relevant surface friction profile for silicone anatomical models.
Echogenic Treatment
BDC Laboratories’ echogenic treatment alters the properties of silicone vessels to more accurately replicate how real anatomical structures appear under clinical ultrasound imaging.
Calcification
BDC Laboratories can integrate custom calcified lesions into any location with vascular or anatomical models, including coronary arteries and aortic valves.
Extend Your System
Add-ons extend your system’s capabilities. They can be purchased to support specific applications and configurations.
Fluid Handling
BDC Laboratories’ fluid handling systems deliver precise, repeatable flow conditions to support the evaluation, testing, and demonstration of cardiovascular devices and their delivery systems. Designed to simulate physiological hemodynamics, these systems range from the digitally controlled PD-1100 to the portable PD-0500 and PD-0750 platforms.
Accurate Pressure Monitoring
The Statys® Pressure Monitoring System delivers precise, reliable pressure measurements for cardiovascular device evaluation or demonstration. Supporting up to four pressure transducers simultaneously, this system combines dedicated data acquisition hardware with user-friendly software to monitor, chart, and record critical pressure signals with accuracy and consistency.
Multi-Camera Imaging
BDC Laboratories’ imaging system provides an easy, reliable way to capture high-quality visuals for flow model simulations. Configurable with up to four compact USB-compatible cameras, the system includes a tablet-based tech package with our Statys® Video Monitoring software to easily record and review multiple views of device placement in real time.
Anatomical Reference Documents
Documentation presenting the dimensional specifications of select BDC Laboratories anatomies, along with corresponding clinical data reported in published literature. Inner diameters, angles, and radii of curvature of the models are identified by letters denoted on the image, with the specification presented in tabular form. The associated published data are presented in tabular form as well, for direct reference, when applicable. BDC Laboratories’ models with Reference Documentation are consistently within one standard deviation of published literature.
Currently Available for:
- Type III Aortic Arch (S01222-T3)
- Neurovascular Arteries (S01104)
- Lower Extremities (SLOWEXTREM-2R)
Why BDC?
Custom-Designed for Your Application
From first conversation through model delivery, we work closely with your team to create silicone vessels engineered to match your specific anatomy, clinical conditions, and testing or training goals.
Expertise You Can Rely On
With 15+ years in silicone vessel fabrication and 40+ in cardiovascular device evaluation, we design for durability and regulatory alignment as needed.
Realism That Engages and Educates
Our vessels can replicate the feel and performance of clinical use, with precise anatomy that make them ideal for hands-on physician training and sales demonstrations.
Explore More

Testing Equipment Brochure

Testing Services Brochure
Silicone Vessels Brochure

Simulation Solutions Brochure
Connect with an Expert at BDC
Provide a few details about your project, and a BDC Laboratories team member will respond within 2 business days to advise on next steps.

