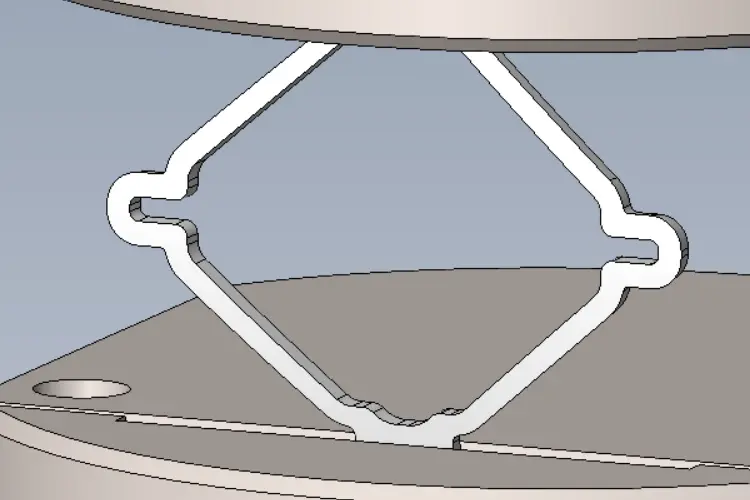
Material Fatigue Strength Testing
Accurate stress-life (S-N) material characterization is critical for the reliable fatigue analysis of medical devices using Finite Element Analysis (FEA). An implant’s specific geometry and manufacturing processes significantly influence S-N fatigue performance, so regulatory bodies often require material fatigue testing on coupon samples that replicate the actual implant or represent its design.
BDC Laboratories works closely with FEA experts to validate the computational models associated with coupon fatigue test samples and to plan the required test load levels for statistics-based S-N fatigue characterization.
Service Highlights
Standards-Driven Excellence
BDC Laboratories’ comprehensive testing capabilities, per the respective published guidance documents, appear below.
Complete Testing Solution
Our team supports all aspects of medical device assessment, including developing the testing strategy, establishing protocols, performing data analyses, and providing regulatory quality final reports.
Scalable Test Program
We execute all test programs according to the data’s application, ranging from abbreviated early-stage development studies to final, formal regulatory submission studies.
Submission-Ready Data
Get tolerance intervals associated with the S-N fatigue test data in comprehensive study reports formatted for regulatory submission.
Relevant Standards
ISO Standards
- 5840-1 (Annex K), Cardiovascular implants — Cardiac valve prostheses — Part 1: General requirements
- 5910, Cardiovascular implants and extracorporeal systems — Cardiac valve repair devices
ASTM Standards
- F3211: Standard Guide for Fatigue-to-Fracture (FtF) Methodology for Cardiovascular Medical Devices
- E739: Standard Practice for Statistical Analysis of Linear or Linearized Stress-Life (S-N) and Strain-Life (ε-N) Fatigue Data

Testing Backed by ISO/IEC 17025 Accreditation
At BDC Laboratories, quality is central to everything we do. We maintain ISO/IEC 17025:2017 accreditation and comply with relevant portions of 21 CFR Part 58 (Good Laboratory Practices), among other U.S. and international governing standards. With a comprehensive quality management system, we ensure service and product integrity with a focus on regulatory compliance and continuous improvement.
Why BDC?
Built Around Your Goals
From first conversation to final report, we collaborate with your team to design and execute accredited testing programs aligned with your device goals and regulatory requirements.
Regulatory-Ready, On Time
With ISO/IEC 17025:2017 accreditation and 40+ years of industry experience, we deliver precise, regulatory-ready results on timelines that keep your project moving.
Reliable, Responsive Partnership
You’ll work with project managers who stay engaged throughout the process. They’re available for questions, quick to respond, and focused on keeping your program on track.
Explore More

Testing Services

Testing Equipment
Silicone Solutions
Simulation Solutions
Connect with an Expert at BDC
Provide a few details about your project, and a BDC Laboratories team member will respond within 2 business days to advise on next steps.

